Venus Medtech successfully implanted the first VenusP-Valve in its pivotal trial in USA
Recently, Venus Medtech (Hangzhou) Inc. ("Venus Medtech"), announced that its independently developed transcatheter pulmonary valve replacement (TPVR) system, VenusP-Valve, has successfully been implanted in the first critical clinical patient in the United States at the University of Virginia School of Medicine (UVA).

The VenusP-Valve is the first Chinese artificial heart valve to receive approval from the U.S. FDA for clinical trials and also the first Chinese artificial heart valve to be approved for market in Europe. Prior to entering formal clinical trials in the United States, the VenusP-Valve had been authorized multiple times for humanitarian use to treat patients, successfully completing several cases in the United States.
Case review
The clinical trial was jointly completed by Professor Scott Lim and Professor Michael Hainstock of the University of Virginia School of Medicine, along with a multidisciplinary team.
The patient, a 13-year-old girl, had previously undergone pulmonary valve commissurotomy and was readmitted due to severe pulmonary valve regurgitation. Ultrasound indicated severe pulmonary valve regurgitation and severe tricuspid valve regurgitation. After assessment, she underwent transcatheter pulmonary valve replacement (TPVR) using the VenusP-Valve.
The procedure was performed under local anesthesia. The patient remained awake as access was gained via the right femoral vein. Preoperative CT analysis, intraoperative fluoroscopy, and balloon measurements guided the precise selection of the VenusP-Valve P30-30 mm valve. The deployment of the VenusP-Valve was successful, with accurate positioning and excellent morphology. Postoperative angiography confirmed good valve function, no regurgitation or paravalvular leaks, and stable hemodynamics. Significant improvement in pulmonary valve regurgitation was observed.
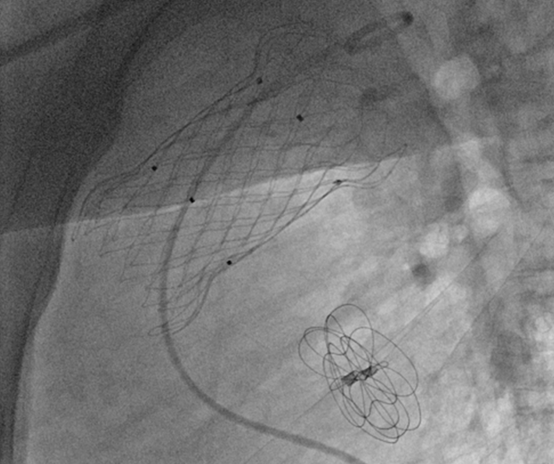
Postoperative imaging

发表留言
暂无留言
输入您的留言参与专家互动